Life sciences solutions, purpose-built for neuropsychiatry
Since 2020, Osmind has supported over a dozen sponsors across top 5 pharma, biotech, and medical device with solutions to develop, launch, and scale breakthrough psychiatric treatments.

The only platform combining EHR, multimodal real-world data, an engaged clinic network, and direct patient access.

Osmind powers the largest network of clinics in the U.S. administering next-generation psychiatric treatments.
Osmind can deploy surveys, micro-surveys, and point-of-care alerts directly within the EHR — enabling primary research, prospective data capture, and recruitment linked to RWD on an identifiable basis, not just passive data extraction.
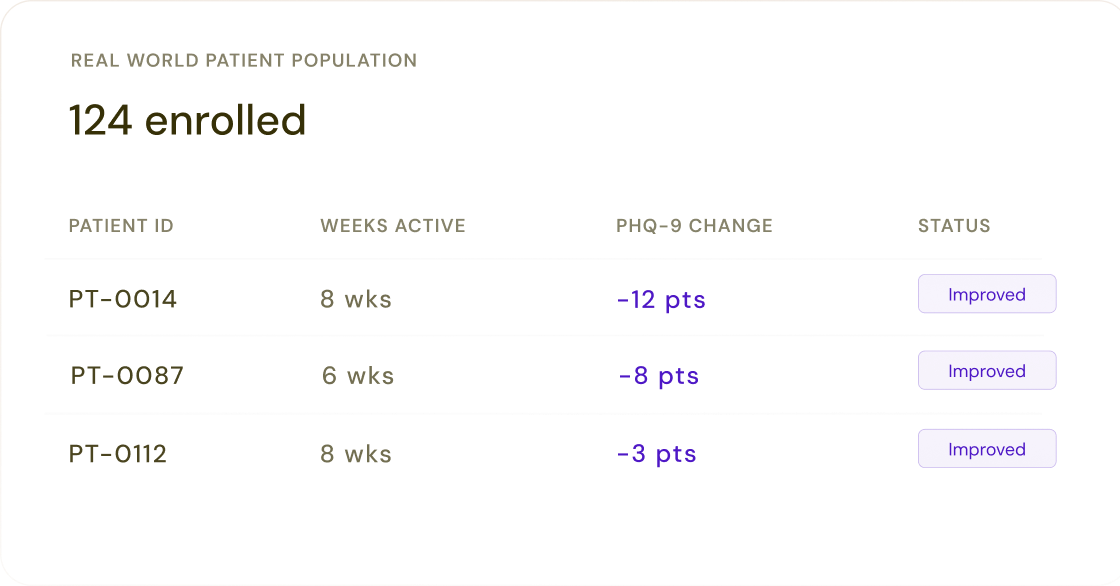
Psychiatric evaluation, medications, outcomes, medical history, dose, route, weight, administration time, and more — captured at the point of care and available immediately, rather than aggregated months later from a data warehouse.
The only psychiatry dataset that directly captures beat-by-beat vitals, audio, video, smartphone, and passive sensing data during and between clinical encounters.
Collected natively in the EHR, with every line item captured and the ability to add fit-for-purpose research variables into the system. Not appended from external sources or imputed from free text.
Tailored solutions for every stage of the development lifecycle
From translational to commercial, Osmind delivers fit-for-purpose solutions. Below are examples from projects with leading sponsors.
.png)
Partnered with a large pharma as principal investigator and recruitment partner for a decentralized trial. Prospectively captured multimodal data to assess domains of depression and identify objective digital biomarkers in real-world patient populations.
Supported a large pharma with data-driven clinical trial design. Leveraged EHR-derived real-world data and machine learning to predict enhanced response to multiple marketed treatments with a similar MOA to their investigational product, translating response enrichment into specific I/E criteria.
.png)
Supported a publicly traded biotech with regulatory submission of a New Drug Application, leveraging proprietary real-world EHR data for supportive evidence across efficacy and safety. Osmind's role spanned regulatory-grade RWD, end-to-end statistical analysis, study design and execution, and direct regulatory support.

Supported a publicly traded biotech with commercial launch planning and forecasting through mixed-methods research. Leveraged real-world EHR and billing/claims data alongside primary research with clinicians and practice managers to detail gaps across coding, reimbursement, clinic economics, and prescribing behavior — directly influencing commercial and market access strategy.

Helped a pharma partner optimize the launch of a novel treatment by addressing barriers to patient identification and treatment inertia. Executed a campaign pairing broad and personalized clinician education with EHR-based in-workflow patient identification tools, alerts, and dashboards that nudged clinicians to take appropriate clinical action.

A pharma partner needed specific data elements unavailable through existing EHR or claims data. Osmind added custom required variables directly into the EHR, creating a fit-for-purpose dataset. An HEOR RWE analysis was initiated after the sample size grew to thousands of patients within weeks.
Bespoke solutions powered by Osmind’s EHR, real-world data, and clinic network
.webp)
Osmind specializes in generating high-depth insights through our unique combination of EHR, RWD, clinician access, and patient access. We strategically combine methods to maximize fit-for-purpose evidence generation:
- Retrospective analysis: EHR, outcomes, billing data
- Prospective data capture: custom scales, clinician-administered scales, digital biomarkers, labs, EEG, genetic testing, etc.
- Physician-led chart review by the same treating physician into fit-for-purpose eCRF
- Clinician survey
- Clinician interview
- NLP / computational processing or expert abstraction of free text clinical notes
- Direct patient survey
- Implementation science study design
.webp)
Leveraging our differentiated combination of point-of-care technology, real-time data, clinic network, and patient pool, Osmind provides next-generation clinical research solutions, including:
- Prospective observational studies and patient registries
- Pragmatic trials and decentralized trials
- EHR and data-driven trials recruitment
.webp)
Most vendors offer one or the other. Osmind is the only platform in neuropsychiatry that can link what clinicians say with what they do — because we see both.
Osmind brings together unmatched subject matter expertise, especially in interventional psychiatry, with a structural data advantage no general-purpose vendor can replicate — giving our partners both the evidence and the context to act on it.
Osmind’s proprietary data and access allows us to be very granular and precise in targeting practices for insight generation, using criteria such as clinical practice, patient populations, and practice infrastructure. Following pre-screening, Osmind leverages our extensive in-house network and position as clinics’ system of record to ensure rapid recruitment timelines. Linking primary research data with EHR data results in highly differentiated, actionable evidence.
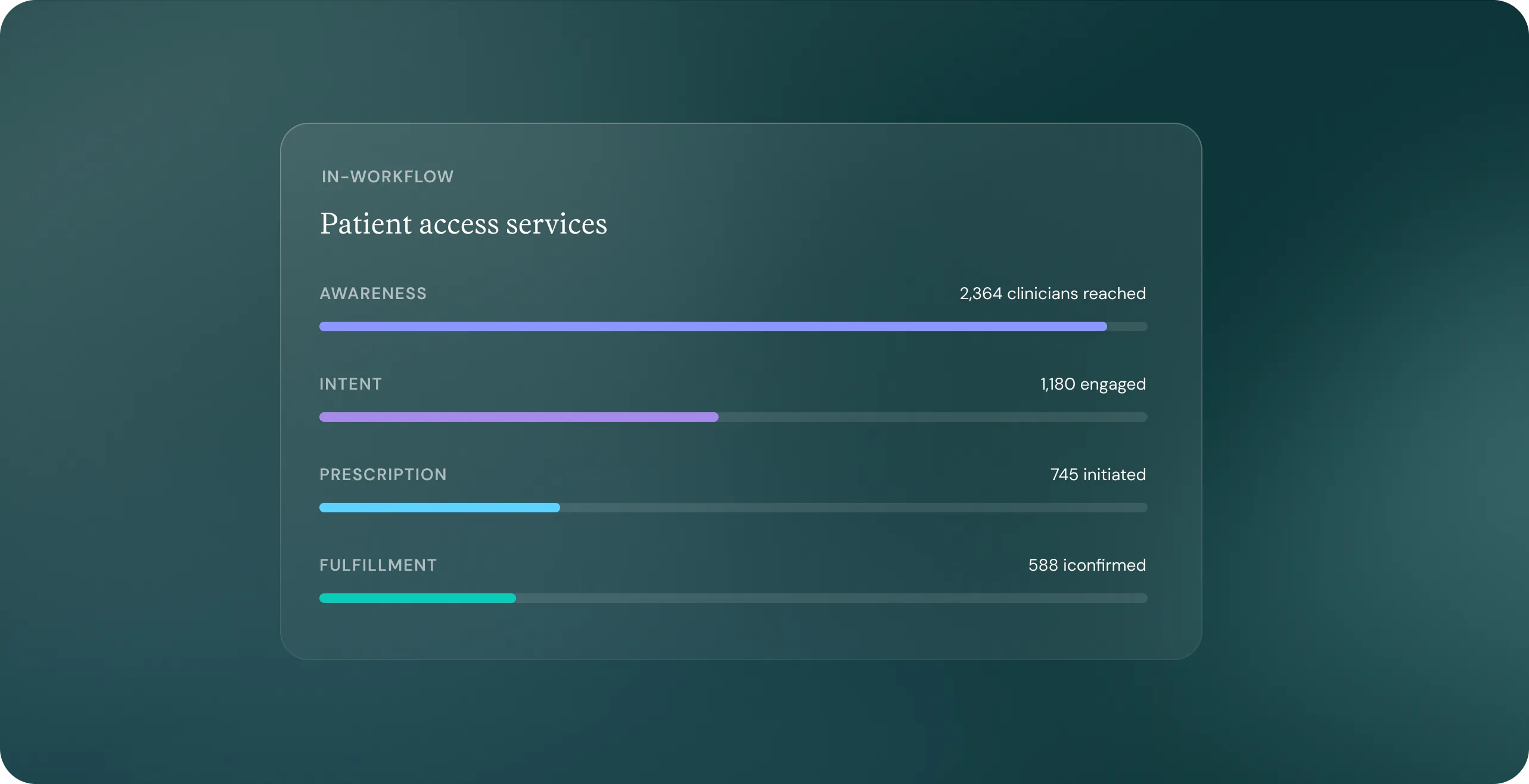
As the technology system of record that powers psychiatry practices, in addition to being the clinic’s go-to partner across billing, revenue cycle management, service line expansion, group purchasing, practice development, and more, Osmind is uniquely positioned to support life sciences with bespoke commercial and marketing solutions.
Osmind has supported brands across the treatment funnel, driving results across awareness, intent, prescription, and fulfillment. We support better patient care by enhancing patient access, minimizing time-to-treatment and maximizing dispense yield.
- Clinician education
- Clinic and practice education and operational support
- In-workflow patient identification
- Patient access services (e.g. reimbursement support, verification of benefits, prior authorizations, drug channel navigation)
- Integration with sponsor services such as REMS and patient hubs
Life Sciences Updates
Osmind Collaborates with Compass Pathways to Advance Independent Clinic Readiness for Psychedelic Treatments
Osmind and CareNet GPO Partner to Make Next-Gen Psychiatric Treatments Accessible and Sustainable to Independent Psychiatry Practices Nationwide
Osmind Supports Successful FDA Type C Meeting in Support of New Drug Application
Osmind partners with LivaNova to Support Education and Awareness for Clinicians Caring for People Living with Difficult-to-treat Depression
Bendable Therapy and Osmind to Present First Real-World Outcomes Data from Legal U.S. Psilocybin Services
Cybin Partners with Osmind to Accelerate Commercial Preparation for its Clinical-Stage Psychiatry Programs
Research & Publications
Explore Osmind’s peer-reviewed publications
Learn more about Osmind’s life sciences solutions
If you, or someone you know, is in crisis or needs immediate assistance, please call 911 immediately. To talk to someone now, please call the National Suicide Prevention Lifeline at 1-800-273-8255 or 988.
Osmind © 2026 All Rights Reserved.
.webp)
.webp)

.webp)